Class IIa medical devices are among the most common products on the European market. This article explains which products fall into this class, which classification rules are relevant, and what regulatory consequences this has.
- The MDR assigns Class IIa to medium risk medical devices.
- This classification has little influence on the legal requirements for the medical device.
- However, the classification does affect the conformity assessment procedure and the intensity of monitoring by notified bodies.
1. Definition: What is a Class IIa medical device?
Class IIa medical devices are products with a medium risk potential according to the EU Medical Device Regulation (MDR) 2017/745. They require the involvement of a notified body in the conformity assessment procedure. Typical examples are diagnostic devices, short-term invasive products, or active therapeutic products for energy delivery, provided that this energy does not pose a potential hazard.
Classification in the classification system The MDR distinguishes between four risk classes:
| Class | Risk potential | Examples |
| I | Low | Bandages, crutches |
| II | Medium | Blood pressure monitors, disposable catheters |
| IIb | Increased | Ventilators, X-ray machines |
| III | High | Pacemakers, hip implants |
Important: Classification is based on the manufacturer’s intended purpose, not on technical complexity or price.
2. Examples: Which products belong to class IIa?
Typical Class IIa products by category
| Category | Examples |
| Diagnostic devices | Blood pressure monitors, clinical thermometers |
| Short-term invasive products | Disposable catheters (< 60 min.), cannulas, tracheal tubes |
| Active therapeutic devices | TENS devices, ultrasound therapy devices |
| Contact lenses | Daily disposable lenses, soft contact lenses (not long-term) |
| Wound care | Wound dressings (non-absorbable, not for chronic wounds) |
| Software | DiGA for therapy support (without diagnosis of serious illnesses) |
The same product category may be classified differently depending on its intended use.
Example: contact lenses
- Daily disposable lenses → Class IIa
- Long-term contact lenses (> 30 days) → Class IIb
3. Classification rules: Which rules lead to Class IIa?
Classification is based on Annex VIII of the MDR. The following rules can lead to Class IIa:
Overview of relevant rules
| Rule | Product type | Class IIa if… |
| 5 | Non-invasive | Intended for the storage/transport of blood, body fluids, or tissue |
| 6 | Non-invasive | Intended to alter the composition of blood/body fluids by filtration, centrifugation, or gas/heat exchange |
| 7 | Invasive (body orifices) | Intended for short-term use (< 60 min) OR long-term use in the oral cavity/ear canal/nasal cavity |
| 9 | Active therapeutic | Energy is delivered in a non-potentially dangerous manner |
| 10 | Actively diagnostic | Intended for direct diagnosis/monitoring of vital bodily functions (without immediate danger) |
| 11 | Software | Information for therapy decisions, if errors do not cause serious deterioration |
| 12 | Drug administration | Administration is carried out in a manner that is not potentially dangerous. |
The classification depends on the individual case. For example, gauze bandages in contact with injured skin are classified higher, there are different types of catheters, and a central venous catheter is a Class III product.
Special feature: Rule 11 (software as a medical device)
Software is classified according to Rule 11. Classification depends on
- the type of information provided,
- decisions based on it, and
- the consequences of misinformation.
Rule 11 in detail: The classification of software is complex and is expected to be adjusted by the planned MDR revision.
→ A detailed description can be found in the technical article Rule 11 – Software as a medical device.
4. Decision tree: Is my product Class IIa?
The following decision tree guides you through the most important classification paths:
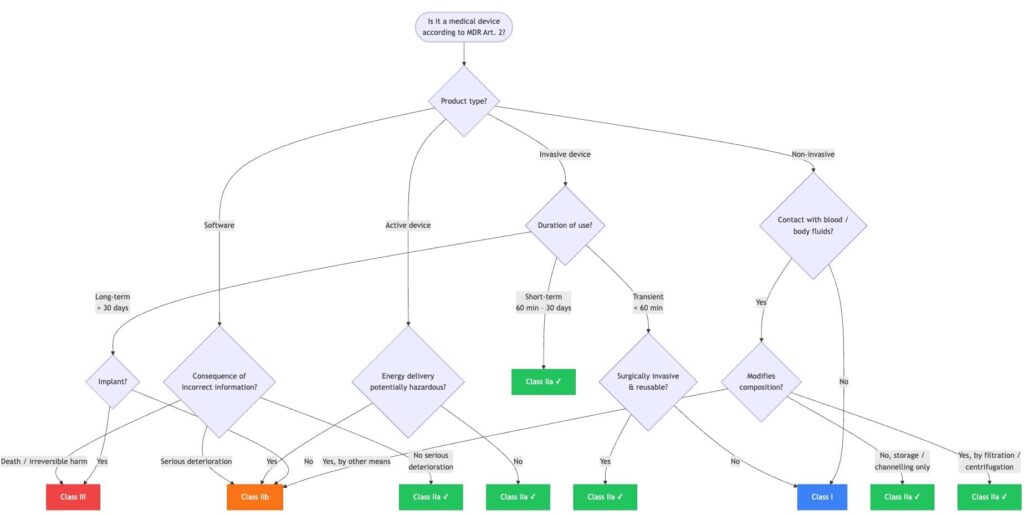
This decision tree covers the most common cases. However, it does not fully reflect all 22 classification rules. In case of doubt, a detailed review based on Annex VIII MDR is required.
5. Consequences: What does Class IIa mean in concrete terms?
The classification has a direct impact on the regulatory burden: a. Requirements in the “class comparison”
| Aspect | Class I | Class IIa | Class IIb | Class III |
| Notified body required | No | Yes | Yes | Yes |
| Conformity assessment | Annex IV | Annex IX (Chapters I+III) or XI Section 10 or Section 18 | Annex IX (Chapters I+III) or X+XI | Annex IX or X+XI Part A |
| TD testing by notified body | No | Random sampling | Random sampling | Complete |
| PSUR frequency | Not required | At least every 2 years | At least annually | At least annually |
| Clinical examination | Rarely | Mostly no | More frequent | Often required |
| Expert panel | No | No | Only implantable | Yes (new products) |
| Instructions for use | Can be omitted** | Can be omitted** | Always required | Always required |
| PRRC required | Yes | Yes | Yes | Yes |
| UDI requirement | Yes | Yes | Yes | Yes |
| EUDAMED registration | Yes | Yes | Yes | Yes |
*Exception: Classes Is, Im, Ir require a notified body
**If safe use is guaranteed without instructions for use (Annex I, Section 23.1)
b. Conformity assessment procedures for Class IIa
There are two options for Class IIa:
- Annex IX, Chapters I + III: QMS assessment + TD testing on a random basis
- Annex XI, Section 10 or Section 18: Product testing
Details on the conformity assessment procedures can be found in our technical article on conformity assessment according to MDR.
c. Sampling of technical documentation
For Class IIa, the notified body does not check every piece of technical documentation in full. Instead, a risk-based sample selection is carried out:
Selection criteria:
- Product categories ( Generic Device Groups )
- At least one product per category
- New products are prioritized
- Products with incidents are prioritized
- Sample size increases with portfolio size
Monitoring audits: Additional samples are checked during annual audits.
Even if your TD is not reviewed, it must be complete and compliant. The Notified Body may carry out additional reviews at any time.
d. PSUR: Periodic Safety Update Report
Manufacturers of Class IIa devices must prepare a PSUR.
- Frequency: At least every 2 years (for Class IIb/III: annually)
- Content: Summary of all PMS data, benefit-risk assessment
- Template: from the Notified Body
For details on PSUR, please refer to our technical article on post-market surveillance and PSUR.
6. What does the classification not affect?
Many requirements apply regardless of class:
| Requirement | Applies to all classes? |
| General safety and performance requirements (Annex I) | ✓ Yes |
| Risk management according to ISO 14971 | ✓ Yes |
| Quality management system | ✓ Yes |
| Post-market surveillance (basic obligation) | ✓ Yes |
| Technical documentation (structure according to Annex II/III) | ✓ Yes |
| Vigilance reporting obligations | ✓ Yes |
| Product liability | ✓ Yes |
Conclusion for manufacturers
The classification primarily determines the scope of the external audit by the notified body – not the internal quality effort. Even Class I products require a complete QMS and compliant technical documentation.
7. International perspective
Does Class IIa also apply outside the EU?
No. The MDR classification only applies in the European Economic Area (EU + Iceland, Liechtenstein, Norway) and Switzerland. Other markets have their own systems:
| Market | System | Comparability |
| USA (FDA) | Class I, II, III | Not directly comparable |
| China (NMPA) | Class I, II, III | Own rules |
| Japan (PMDA) | Class I–IV | Four classes |
Example: A Class IIa product in the EU may be Class II or Class III in the US.
→ For international approval strategies: Advice on regulatory strategy
8. Transition periods: What applies to existing products?
Some products are classified higher under the MDR than under the previous MDD:
Examples of upgrades:
- Reusable surgical instruments: I → Ir (with notified body)
- Certain software: I → IIa or higher
- Central venous catheters for short-term use: IIa → III
Details on transition periods and legacy devices can be found in this technical article on MDR transition periods.
9. Can the classification be influenced?
The classification is based on the intended purpose, which is determined by the manufacturer. Legitimate strategies
| Strategy | Example |
| Limit the range of functions | Software only displays instead of analyzing → lower class |
| Limit duration of use | Short-term instead of long-term invasive → lower class |
| Specify indication | Exclusion of certain patient groups |
| Product division | Separation into several products with different classes |
- Regulatory problems (audits, market surveillance)
- Liability risks
- Reputational damage
10. Summary
Class IIa medical devices:
- These are products with medium risk potential.
- The involvement of a notified body is required.
- The technical documentation is reviewed on a random basis.
- A PSUR must be prepared at least every 2 years.
- The preparation of instructions for use may not be necessary in some circumstances.
Most important classification rules: 5, 6, 7, 9, 10, 11, 12
Crucial: The intended purpose determines the class – not the technology.
Are you unsure about the classification of your product? In our “Regulatory Strategy” consultation, we support you with:
- Classification according to MDR (and internationally)
- Qualification as a medical device vs. non-medical device
- Optimization of the intended purpose
- Developing a global approval strategy
Dear Ms. Katharina Keutgen,
The recent article “Class IIa Medical Devices: Definition, Examples, and Requirements” provided extremely valuable information, particularly Figure 1 in “4. Decision Tree: Is My Product Class IIa?” which was particularly clear and helpful. Vielen Dank.
Regarding emergency bandages, which are classified as Class 1 in Japan and the U.S., how would they be categorized in the EU? These bandages contain no active pharmaceutical ingredients and are strictly designed for basic wound coverage and protection purposes.
According to Figure 1, they are non-invasive, have no contact with blood or bodily fluids, and are classified as Class 1.
Is this understanding correct?
Regulatory Science Consultant
Hiroshi Uno
Dear Hiroshi Uno,
thank you very much for your interesting question.
From Figure 1 you are right; however, this decision tree is simplified and covers the most common cases but it does not fully reflect all 22 classification rules.
The classification depends on the intended use of the device. For the emergency bandages most probably Rule 4 of Annex VIII is relevant:
“All non-invasive devices which come into contact with injured skin or mucous membrane are classified as:
— class I if they are intended to be used as a mechanical barrier, for compression or for absorption of exudates;
— class IIb if they are intended to be used principally for injuries to skin which have breached the dermis or mucous membrane and can only heal by secondary intent;
— class IIa if they are principally intended to manage the micro-environment of injured skin or mucous membrane; and
— class IIa in all other cases.
This rule applies also to the invasive devices that come into contact with injured mucous membrane.”
Following your description, the bandages would be class I devices. Please note, that for sterile class I devices, a notified body has to be involved in the conformity assessment procedure nevertheless.
Dear Katharina Keutgen,
I sincerely appreciate your prompt and courteous response.
After reviewing MDR Annex VIII, Section 4. NON-INVASIVE DEVICES again,
I understood that Rule 4 would be the most appropriate regulation for our interest in emergency bandages.
I also believe the unsterilized nature of the product would not constitute a Class 1 deviation.
With this, I consider this matter closed.
I hope you continue to encounter even more favorable business opportunities in the future.
Best regards,
Hiroshi Uno