Medical device and IVD manufacturers must distinguish the concepts of “abnormal use,” “foreseeable misuse,” “errors in normal use,” and “misuse.” This is a prerequisite for understanding and complying with ISO 14971 and IEC 62366 requirements.
Even the measures that manufacturers must take depend on the type of use.
1. Definitions and examples
1.1 Normal use
According to ISO 14971, intended purpose/use refers to the
use for which a product, process or service is intended according to the specifications, instructions and information provided by the manufacturer
The article on intended purpose and intended use compares further definitions of the term.
According to the understanding of IEC 62366-1, users can make mistakes even when using the device as intended. That may be counterintuitive for some.
1.2 Abnormal use
Conscious, intentional act or intentional omission of an act that is counter to or violates normal use and is also beyond any further reasonable means of user interface-related risk control by the manufacturer.
IEC 62366-1:2021, 3.1
Use is thus abnormal if three conditions are met:
- The user acts consciously or consciously refrains from acting.
- The user infringes/violates normal use.
- The manufacturer can hardly control the risks associated with this.
Why this controllability has been included in this definition remains a secret of the standards committees. It may be that ultimate risk control is meant because under certain circumstances, the user or the organization that purchased the medical device is ultimately responsible for its use.
The standard cites the following as examples of abnormal use:
- Grossly negligent use
- Sabotage
- Intentional disregard of safety information
Abnormal use would be when a hospital technician disables the safety alarms on a respirator to prevent them from being “distractedly triggered.”
1.3 Foreseeable misuse
ISO 14971 requires manufacturers to “document reasonably foreseeable misuse.” It now provides a definition of “reasonably foreseeable misuse.”
use of a product or system in a way not intended by the manufacturer, but which can result from readily predictable human behaviour
DIN EN ISO 14971:2022 3.15
The standard makes it clear that this misuse can be intentional or unintentional.
Almost the same definition is given in DIN EN 82079-1.
use of a product or system in a way not intended by the supplier, but which can result from readily predictable human behaviour
There is no clear-cut distinction between predictable and unpredictable. The probability of this action is crucial.
If an action has occurred once, an action would have to be classified as “reasonably foreseeable.”
Post-market surveillance (market surveillance) also helps to identify such actions as they are documented.
An example of “foreseeable misuse” would be a patient who uses the same blood glucose meter but shares the lancet for taking blood samples with other family members, even though the instructions for use explicitly state that the lancets are for single use only.
An example of “unforeseeable misuse” would be a surgeon using a disposable scalpel designed for use in general surgery for experimental micro-neurosurgical procedures for which it was neither designed nor suitable.
This application is so far outside the intended use and is so unusual that the manufacturer could not reasonably have foreseen it.
1.4 Short version
The standards IEC 62366-1 and ISO 14971 work with different taxonomies.
1.4.1 IEC 62366-1 taxonomy
IEC 62366-1 initially differentiates between normal use and abnormal use.
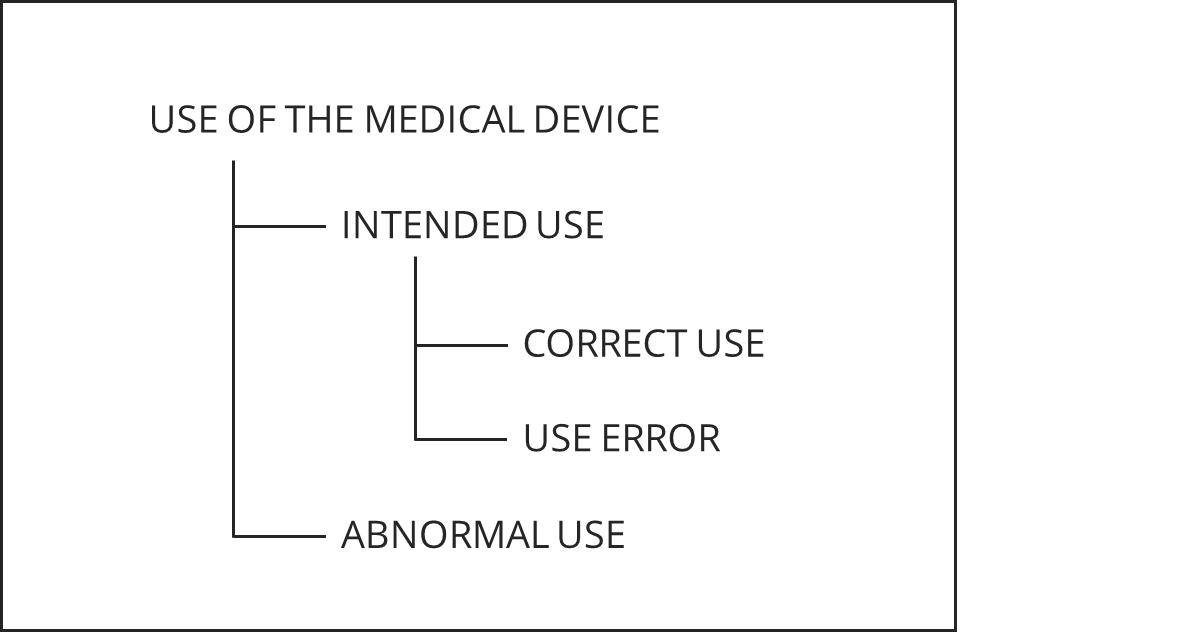
IEC 62366 does not distinguish “normatively” between foreseeable and unforeseeable misuse. However, Annex D states that no standard can address unforeseeable actions.
IEC 62366-1:2015 no longer recognizes the taxonomy of errors from the previous standard (see Fig. B.1). One reason is that the distinctions between the taxonomy classes were not always clear and helpful. For example, whether a user enters a laboratory value in mmol/l instead of the expected mg/dl because he did not look properly (an error of attention) or because he was unaware of the difference (an error) often led to discussions. Admittedly, the risk-minimizing measures may differ.
1.4.2 ISO 14971 taxonomy
On the other hand, ISO 14971 does distinguish between reasonably foreseeable and not reasonably foreseeable misuse. However, it does not recognize the concept of abnormal use.
2. Regulatory framework
2.1 Scope of the standards
Neither ISO 14971 nor IEC 62366-1 require consideration of reasonably unforeseeable misuse.
Abnormal use is outside the scope of IEC 62366-1. If the abnormal use is foreseeable, it falls within the scope of ISO 14971.
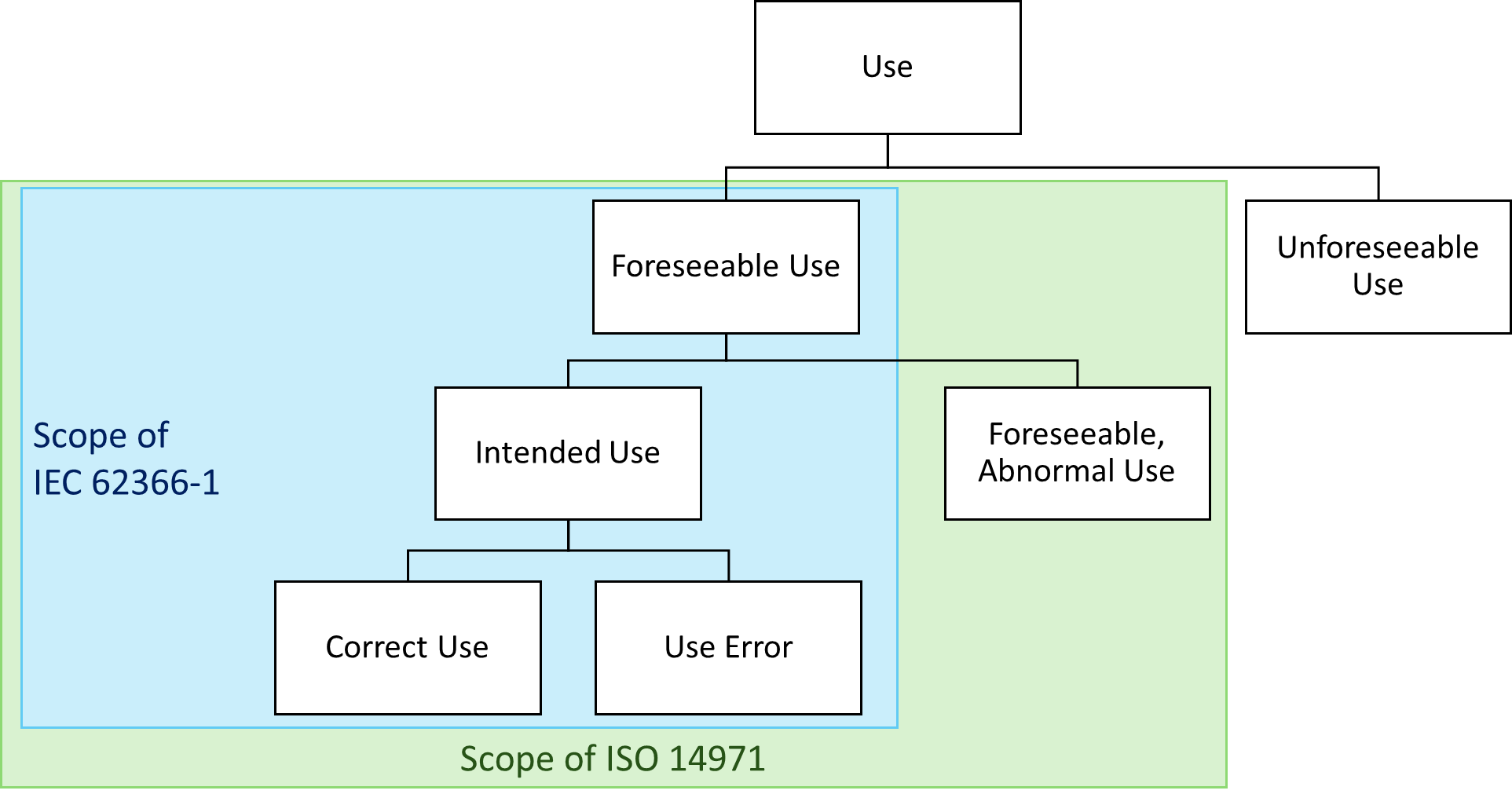
The above-described reuse of the lancet must be considered in risk management according to ISO 14971. However, it is not the subject of IEC 62366-1. Thus, there is no obligation to describe a corresponding “use scenario.”
ISO 14971 also considers the “bad user” who could misuse the medical device (predictably!). Such misuse could consist of the user using it for an unforeseen purpose, consciously ignoring safety instructions, or using it without appropriate training, although this is explicitly required.
IEC 62366, on the other hand, assumes the “good user.” It only considers errors that arise because the user forgets something, is inattentive, or makes a mistake.
In the case of an error, the user acts with full (and good) intention but endangers a patient or user. For example, this could happen because the user misunderstood the device. The standard refers to a “wrong mental model.”
Fig. 2 shows the foreseeable misuse in the boxes “use errors” and “foreseeable abnormal use.”
2.2 MDR and IVDR requirements
MDR and IVDR require manufacturers to minimize the risk of use errors (Annex I, Section 5). They also oblige manufacturers to carry out post-market surveillance. Part of this post-market surveillance should concern the use of the devices.
2.3 ISO 14971 requirements
The international standard for risk management (ISO 14971) requires that a manufacturer document not only the intended purpose but also the reasonably foreseeable misuse. Both must be considered in the risk analysis and appropriate measures taken.
The reasonably unforeseeable misuse does not have to be considered. Nevertheless, the manufacturer can take action via protective mechanisms and IFU warnings.
2.4 IEC 62366-1 requirements
IEC 62366-1 requires manufacturers to identify usability risks, take measures to address them, and evaluate the effectiveness of those measures in formative and summative evaluations.
2.5 IEC 60601-1 requirements
Manufacturers must ensure that medical devices fulfill the applicable requirements of IEC 60601-1, even in the event of foreseeable misuse.
3. Summary and conclusion
3.1 Counterintuitive and imprecise definitions
Not all of the terms considered here are intuitive.
- Many people understand misuse to mean acting with bad intentions, but that is not consistent with the definitions.
- The foreseeability of an action has nothing to do with whether it is committed intentionally or unintentionally. The conceptual and interpretational worlds of the two standards collide here, and they are not well aligned.
- Many people equate “conceivable” with “foreseeable.” However, IEC 62366-1 only recognizes “foreseeable.”
- Even if a “use error” is made, normal use can still be present.
3.2 Unforeseeable or not foreseen?
One can agree with the statement in IEC 62366-1:2015 that a standard cannot deal with unforeseeable actions. However, a clear distinction should be made between not anticipating something and dismissing something as unforeseeable.
3.3 Conclusion
Precise and uniformly defined terms in all relevant standards are necessary to avoid discussions and to create precise documentation. If explanatory comments are necessary due to the complexity of the relationships between the terms, this should not be done by means of so-called “notes”, but rather, if possible, by means of memorable examples in the informative annexes. The standards still have some catching up to do here.
Change history:
- 2025-03-25: This new article replaces two obsolete articles on “foreseeable misuse” and “abnormal use”
What about OFF-LABEL use? Is this term reserved for medications only, or can be used also for medical devices?
Dear Irena,
Thank you for this important question! I have not read this term in official documents for medical devices. However, I assume that it can also be transferred from the pharmaceutical world to medical devices. Off-label use is often the case when a doctor deliberately wants to use a medicine (or medical device) for a target patient group that is not part of the intended use. Doctors then take responsibility on the grounds that not using the product would entail much greater risks than using it.
From the point of view of ISO 14971, this would also be an abnormal use within the scope of reasonably foreseeable misuse. So, it is a very specific type of “reasonably foreseeable misuse”.
Best regards
Christian
While ISO14971 is actually quite vague on on this topic (reasonably foreseeable misuse), the risk guidance ISO/TR 24971 §5.2 (Intended use and reasonably foreseeable misuse) provides better clarity; explicitly addressing ‘abnormal use’ in the context of ‘reasonably foreseeable misuse’ with examples.
Thanks Jonas for the valuable supplement!
In fact, ISO 24971 provides all the necessary background information for implementing ISO 14971, as the latter only contains the core requirements.